 Problem7 How many valence electrons do Fluorine have? Because when you look at the electronic configuration of oxygen, there are 6 electrons present in the last cell(valence cell). 
The valence electron of oxygen will be 6. Problem6 How many valence electrons does oxygen have? There are also many elements that have more than one valence. Iron will lose two electrons at some point and turn into rt and at another time it will lose three electrons and turn into py. Problem5 Give examples of elements that have more than one valence. The valence of the hydrogen and the valence electron will both be equal. Problem4 Give examples of elements other than carbon whose valency and valence electrons will be equal. Thus, the valency electron of oxide ion is 8. If you look at the electronic configuration of oxide ions, you can see that there are 8 electrons present in the valence cell. Problem3 What is the valency electron of oxide ion? And the last cell of neon will always have 8 electrons. 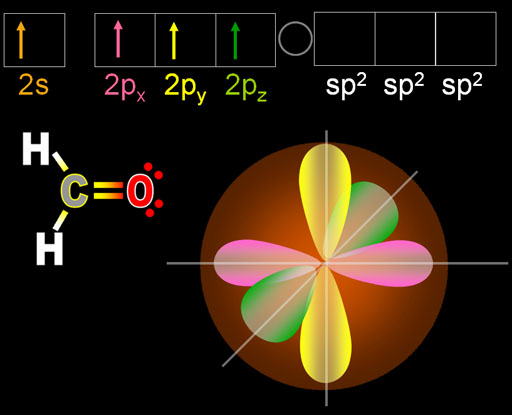
In this case, the valency of neon will be zero. That is, the neon will not receive or reject any electrons. Problem2 What is the electron valency of neon and how many electrons are present in its last cell? Will be equal in the case of those elements whose number of electrons in the last cell will be equal to the number of electrons received or rejected for the stability of that element. Valence electrons have no relation with Valency. So will valence and valence electrons be equal or different in case of all other elements? Problem1 In the case of carbon, valency, and valence electrons are equal. 
The question-and-answer on valence and valence electrons is fully discussed below. Question Answer on valency and valence electrons For this, carbon always forms covalent bonds by mutually sharing its own 4 electrons. Because in this case, you have to give a lot of energy. Because it never becomes a c -4or c +4ion by accepting or rejecting the 4 electrons in the last cell in order to gain its stability. 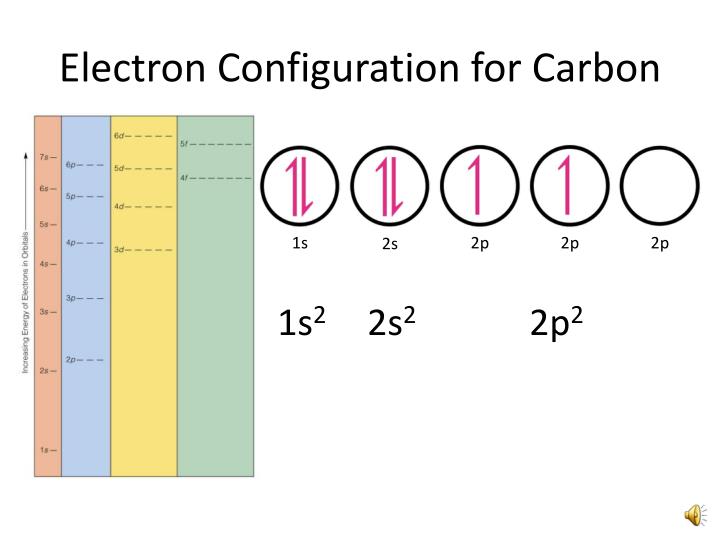
In order for this element to be stable, the electrons in its last cell have to be mutually shared with other elements.Īn example is a carbon. There are some elements that can neither gain nor lose electrons in their last cell. Many of you may be confused as to where the word mutual share in definition came from. Thus, the valency of carbon is four.Īfter each element gains stability, its electronic configuration will be like the nearest Nobel gas. What is Valancy of CarbonĪccording to the octet theory, in order for each element to reach a stable state, the number of electrons it leaves, or gain or mutual shares in order to fill its octet, we will call that element the valence. So, for this, you need to understand the difference between valency and valence electrons. So look at this illustration of the electric configuration of carbon.Īlso, many of you are confused between the valency of carbon and valence electron. The first cell has two electrons and the last cell has four electrons. If you look at the electronic configuration of carbon, you can see that there are two cells present in carbon. That is, there are 4 electrons present in the last cell of carbon. The above two questions mean the same thing, so the carbon has four valence electrons. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
